What Type of Insulin Does Continues Subcutaneous Therapy Use
Identification of relevant studies
The initial search using the search strategy described above yielded 1,228 references. After exclusion of duplicates, there were 673 hits, of which 557 were classified as irrelevant by the reviewers on the basis of their abstracts. Among the remaining 116 publications that were perused in full text, 83 did not meet the inclusion criteria. For further detailed information see Fig. 1. No further full published study was obtained by screening the references of the 109 secondary literature publications retrieved. In the end, 33 publications with usable information were included in our systematic review. Of these, 17 studies (27 publications [13–39]) included mainly adult type 1 diabetic patients, three trials (four publications [40–43]) only children of up to 18 years and two studies (two publications [44, 45]) examined patients with type 2 diabetes mellitus only.
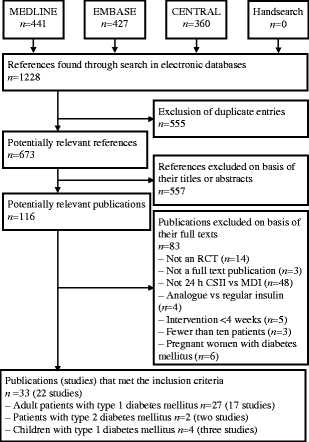
Flow diagram of the literature search and identification of the studies relevant for inclusion in the systematic review. RCT, randomised controlled trial
Description of included trials
Adult patients with type 1 diabetes mellitus
All studies had an open-label study design. Seven of the included studies were parallel studies and ten had a crossover design. Altogether 908 participants took part in the 17 randomised controlled studies. The study periods lasted from a minimum of 5 weeks to a maximum of 2 years. Upon our request, the authors of the Oslo Study [17, 18, 23, 25–29] told us that the study was originally intended to last for 2 years, but was later extended to 4 years. Due to incomplete data records after 4 years, only the results from the 2 year analysis were considered for this review. Twelve studies used regular human or porcine insulin and five studies used insulin analogues as short-acting insulin for CSII and MDI treatment. For further detailed information on the type of insulin and the type of pumps used, see Table 1. For further information on special study characteristics, such as indications for CSII treatment, injection rates and other relevant aspects of care, see ESM Table 2.
In one study [32], MDI treatment was compared with two different CSII regimens, one with insulin pump therapy with a basal fixed overnight insulin rate, the other with insulin pump therapy with a higher variable dawn insulin rate (0500 to 0800 hours). The quality of 15 studies was assessed to be in category C and only two studies were of higher quality (category B). One study had such a serious quality deficiency [32] that we did not include the study results in the respective analyses. In that study, patients were asked immediately after a satisfactory randomisation process (n= 96 patients) if they accepted their assigned treatment. If they refused, they were excluded from the study. This is not standard procedure in trials, and in fact 32 patients in this study dropped out before even starting with the assigned treatment.
Patients with type 2 diabetes mellitus
Both studies had an open-label parallel study design [44, 45] and 234 patients took part in the randomised controlled trials. The study periods were 24 and 52 weeks, respectively. Both studies used insulin analogues as short-acting insulin for CSII and MDI treatment. For further detailed information on the type of insulin and the type of pumps used, see Table 1. The quality of one study was assessed to be in category C and the other study was of higher quality (category B).
Children with type 1 diabetes mellitus
Of the randomised controlled trials found, one [41] used a crossover design, whereas the other two, far more recent investigations [40, 43], were of parallel design. Altogether, 74 children took part in the three randomised controlled studies. The duration of follow-up and treatment was 16 weeks in two trials and 52 weeks in the third trial. Two studies were performed in adolescents [40, 41]. One study [43] included young children with a mean age of 4 years. In the older study [41], regular insulin was used; in both recent studies [40, 43] insulin analogues were used as pump insulin and as bolus insulin in the respective comparison groups. Insulin glargine was used as basal insulin in the MDI groups by all patients in one trial [40] and by 60% of patients at the end of the other study [43], while the other patients in that trial used NPH and ultralente as basal insulin. The quality of one study was assessed to be in category C and the other two studies were of higher quality (category B).
Glycaemic control—glycated haemoglobin
Adult patients with type 1 diabetes mellitus—meta-analysis
Of 17 selected studies we had to exclude five studies from this meta-analysis for the following reasons: (1) one study did not report any HbA1c baseline and follow-up data [13]; (2) one study compared only treatment sequences (CSII-MDI vs MDI-CSII) at study endpoint [24]; (3) another trial provided no measure of variability [37]; (4) in one study [36] the discrepancies in the measurement of variability could not be resolved; and (5) another study [32] had an unacceptably high dropout rate immediately after randomisation assignment.
Ultimately, 12 studies were included in the meta-analysis of the percentage of glycated haemoglobin at the end of treatment. When the SMD was calculated, the effect between treatment groups was estimated to be −0.6 (95% CI: −0.87, −0.22) in favour of CSII compared with MDI (Fig. 2), which corresponds to an effect size of −0.6% in original units. The I 2 statistic was estimated to be 75%, which indicates substantial heterogeneity.
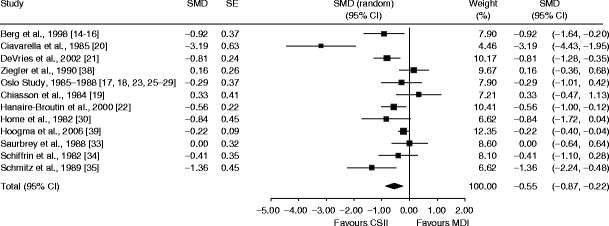
Glycated haemoglobin at the end of treatment in the studies performed on adult patients with type 1 diabetes: standardised mean difference (SMD) between groups. Model with random effects: DerSimonian–Laird. Heterogeneity: Q = 43.16, df = 11 (p = 0.000), I 2 = 74.5%. Overall effect: z score = −3.31 (p = 0.001), τ 2 = 0.212
Six of the twelve studies used HbA1c, whereas the other six studies used HbA1 for the measurement of glycated haemoglobin. Analyses according to the different methods showed that for studies using HbA1c the difference between the CSII and MDI groups was statistically significant in favour of CSII treatment with a WMD of −0.4% (95% CI: −0.65, −0.20; I 2= 72%) (Fig. 3a). For the studies in which HbA1 was measured, the WMD was −0.6% (95% CI: −1.34, 0.14; I 2= 84%). The difference between the groups was not statistically significant (Fig. 3b).
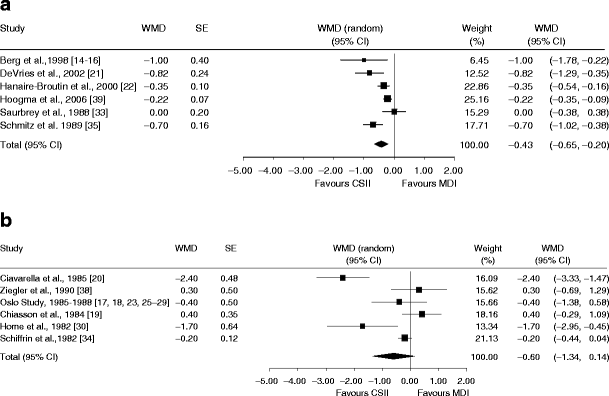
a, b Glycated haemoglobin at the end of treatment in the studies performed on adult patients with type 1 diabetes: weighted mean difference (WMD) between groups analysed by method of measurement. a Studies using glycated HbA1c. Model with random effects: DerSimonian–Laird. Heterogeneity: Q = 17.77, df = 5 (p = 0.003), I 2 = 71.9%. Overall effect: z score = −3.7 (p = 0.000), τ 2 = 0.048. b Studies using glycated HbA1. Model with random effects: DerSimonian–Laird. Heterogeneity: Q = 30.34, df = 5 (p = 0.000), I 2 = 83.5%. Overall effect: z score = −1.59 (p = 0.113), τ 2 = 0.659
In studies with a duration of less than 6 months, the SMD in glycated haemoglobin when comparing CSII with MDI treatment was −0.4 (95% CI: −0.82, −0.01), which is equivalent to −0.4% in original units. In studies of long-term duration (6 months or more) comparing CSII with MDI treatment, the SMD was −0.7 (95% CI: −1.24, −0.19), which corresponds to −0.8% in original units. In both analyses, there was evidence of heterogeneity among short (I 2= 55%) and long-term studies (I 2= 82%). For studies using a parallel design, the SMD in glycated haemoglobin was −0.9 (95% CI: −1.64, −0.10; I 2 = 85%) when comparing CSII and MDI treatment, corresponding to −1.2% in original units. For studies with crossover design the SMD in glycated haemoglobin was −0.4 (95% CI: −0.68, −0.07; I 2 = 52%), corresponding to −0.4% in original units. For studies published in 2000 or later, the WMD was −0.4% (95% CI: −0.60, −0.14; I 2 = 69%) when comparing CSII and MDI treatment.
Patients with type 2 diabetes mellitus
In both trials [44, 45] no significant differences in HbA1c between the two treatment groups were found at the end of the study (Table 1).
Children with type 1 diabetes mellitus
Glycated haemoglobin decreased more pronouncedly in patients treated with CSII in all three included trials. At the end of treatment HbA1c/HbA1 was significantly lower in the CSII group in each of the trials performed in adolescents [40, 41]. In the trial including young children [43], glycated haemoglobin was slightly higher in CSII-treated patients but without reaching statistical significance (Table 1).
Total insulin requirements
Adult patients with type 1 diabetes mellitus
Of 14 studies reporting total insulin requirements at the end of treatment, 12 [13, 19, 22, 24, 28, 30, 33–37, 39] consistently found that the insulin doses were lower in CSII-treated patients. In seven [13, 19, 22, 30, 35, 36, 39] of these investigations, the difference was statistically significant.
One study [20] did not find any difference between the treatment groups and another [16] showed insulin requirements to be higher under pump therapy, but without statistical significance. In the latter trials, patients randomised to CSII treatment started treatment with a higher insulin dosage and could either reduce it more efficiently or showed a smaller increase in required insulin doses during the study period than patients in the MDI groups.
Patients with type 2 diabetes mellitus
There were no reported significant differences in insulin requirements at the end of studies. Raskin et al. [45] showed a slight reduction in insulin requirements in the CSII group compared with the MDI group, while Herman et al. [44] did not find any difference at all.
Children with type 1 diabetes mellitus
At the end of treatment, all three studies reported total insulin requirements to be lower in the CSII group than in the MDI group, which in the two studies with adolescents [40, 41] was statistically significant.
Hypoglycaemia
Divergence between studies
Due to differences in the definitions of hypoglycaemic episodes and in the study duration (5 to 104 weeks), and because of the often missing measures of variance and the small numbers of patients with severe hypoglycaemic episodes, no meta-analysis of hypoglycaemic events was performed.
Severe hypoglycaemia, adult patients with type 1 diabetes mellitus
All in all, severe hypoglycaemic episodes were rare. Four of 17 studies [16, 19, 20, 30] did not mention severe hypoglycaemic episodes. Three others [33, 35, 36] reported that no severe hypoglycaemia was observed. Information on the number of patients with severe hypoglycaemic episodes could be found in six studies [13, 21, 22, 24, 28, 34] and four [32, 37–39] at least reported on the rates or the number of severe hypoglycaemic events in the treatment groups. The proportion of patients with severe hypoglycaemic episodes ranged from 0 to 0.13 in the CSII group and from 0 to 0.4 in the MDI group. Only two studies [21, 28] reported a formal statistical test result for the number of patients with severe hypoglycaemic episodes in the treatment groups, which in both studies did not differ significantly.
Mild/minor/symptomatic hypoglycaemia in adult patients with type 1 diabetes mellitus
In six of 17 studies [13, 16, 19, 20, 33, 35] there were no reports on mild, minor or symptomatic hypoglycaemic events. Two other studies [30, 37] presented results for overall hypoglycaemia only, but for the latter [37] the rate of mild/minor hypoglycaemic events could be calculated because exact numbers of severe events were available. Six studies [21, 22, 24, 28, 36, 39] provided the event rates for mild/minor hypoglycaemia per patient per time period, which ranged from 0.9 to 3.1 weekly events per patient in the CSII groups and from 1.1 to 3.3 in the MDI groups (ranges include previously mentioned study [37]). The median rate per patient per week was 1.9 in the CSII groups and 1.7 in the MDI groups. Due to its high dropout rate one further study [32] that also reported event rates per patient per time was not considered in the above summary. One study [34] presented only the total number of mild and moderate events for treatment. The 2 year trial by Ziegler [38] reported just the rate of symptomatic events per group for 6 month periods; it was always higher in the CSII group and statistically significant in three of these periods.
Severe hypoglycaemia, patients with type 2 diabetes mellitus
Information on severe hypoglycaemia is provided in both publications. All in all, severe hypoglycaemic episodes were rare. In one study [45], there were no severe hypoglycaemic episodes and in the other study [44] three patients in the CSII compared with six patients in the MDI group experienced severe hypoglycaemia. The event rate per patient-year was not significantly different, although it tended towards a reduction in favour of CSII treatment (CSII, 0.1 vs MDI, 0.2 events per patient-year).
Mild hypoglycaemia, patients with type 2 diabetes mellitus
Detailed data for the occurrence of mild hypoglycaemic episodes are given in both studies included here [44, 45], with a non-significant reduction of frequency in the CSII group in the former [44] (CSII, 1.1 vs MDI, 1.2 events per patient per week). In Raskin [45] the event rate was also lower in the CSII group, with 0.8 vs 1.2 events per patient per 30 days in the MDI-treated group. But a formal statistical test was not presented.
Severe hypoglycaemia, children with type 1 diabetes mellitus
In all included trials, the number of severe hypoglycaemic events was small. Only three severe hypoglycaemic events were recorded for patients in the CSII groups and six events in the MDI groups in all trials taken together. In one investigation [40], four patients in the MDI group versus one in the CSII group experienced each exactly one severe hypoglycaemic event. No difference was found for CSII and MDI treatment in the other two trials, with one event in each group in both studies [41, 43], respectively.
No information on mild/minor/symptomatic hypoglycaemia in children with type 1 diabetes mellitus was available.
Adverse events
Adult patients with type 1 diabetes mellitus
Overall, information on adverse treatment effects other than hypoglycaemia was insufficient in the available publications.
Only four studies reported on serious adverse events besides hypoglycaemia. The rates of occurrence were generally low and in two studies [35, 36] there were no serious adverse events. In another study [38] only one serious adverse event was reported as causing a dropout in the CSII group. Another team [39] reported 15 serious adverse events in the MDI group compared with 20 events in the CSII group (including four events of ketoacidosis).
Data on ketoacidosis were given in ten studies. Only one of these [32] reported a statistically significant increase in the occurrence of ketoacidosis in the MDI group compared with both CSII groups (fixed and variable infusion rate), but this study was not considered due to major quality deficiencies after the randomisation process. Ziegler et al. [38] reported data on ketoacidosis indicating that the events per 100 patient-years were higher in the CSII group (intention to treat and per protocol analysis), but the difference was not statistically significant. All the other studies noted only a few ketotic events: in three studies there was one event in the CSII group only [24, 33, 36], another [21] reported one event in both treatment arms, yet another [39] reported four events in the CSII and none in the MDI group and the Oslo trial [28] found that two patients in the CSII vs no patients in the MDI group developed ketoacidosis. Data on hyperglycaemia were given in only one publication included in this review. However, due to major quality deficiencies, the result of hyperglycaemic episodes in that study [32] will not be considered for interpretation.
Four studies reported on infusion site problems, all with an increased rate in the CSII group. Hoogma [39] found that 8.2% of the patients in the CSII vs 0.8% in the MDI group had problems at the infusion site, without giving more detailed information. In another investigation [33], three episodes of subcutaneous infection (one requiring surgical incision) occurred in the same patient in the CSII group, who also had ketoacidosis. Elsewhere [36], infusion site problems were noted under pump therapy only twice and several reasons for infusion problems were reported, i.e. catheter problems, pump arrest or dosage errors, battery problems, syringe dislocation. The Oslo Study [28] reported that eight events in six patients in the CSII vs none in the MDI group involved subcutaneous abscesses. Only one publication [32] included information on mortality, reporting that in the CSII group with variable infusion rates, one patient died. This patient had presented with high blood glucose values the last 2 days before his death, he suffered from fever and acetonuria, and his blood glucose values had not responded to an increased insulin infusion rate.
Patients with type 2 diabetes mellitus
Both publications provided information on local reactions at the injection site. In one [44] significantly more patients in the MDI group reported bleeding or bruising events as compared with the CSII group, where significantly more patients had infusion site inflammation or irritation. In the other study [45] only CSII-treated patients reported injection site reactions, but no data on significance were given. Both teams found that technical problems were more common in the respective CSII groups than in the MDI groups. Data on hyperglycaemic episodes were given in one publication [45] included in this review. These episodes occurred considerably more frequently in MDI patients, but nothing was said about possible statistical significance.
Children with type 1 diabetes mellitus
Doyle [40] reported one episode of ketoacidosis for each of the two treatment groups, while the other study [43] reported no event for any of the groups. There is no more information on any other adverse events in any of the publications.
Source: https://link.springer.com/article/10.1007/s00125-008-0974-3
0 Response to "What Type of Insulin Does Continues Subcutaneous Therapy Use"
Post a Comment